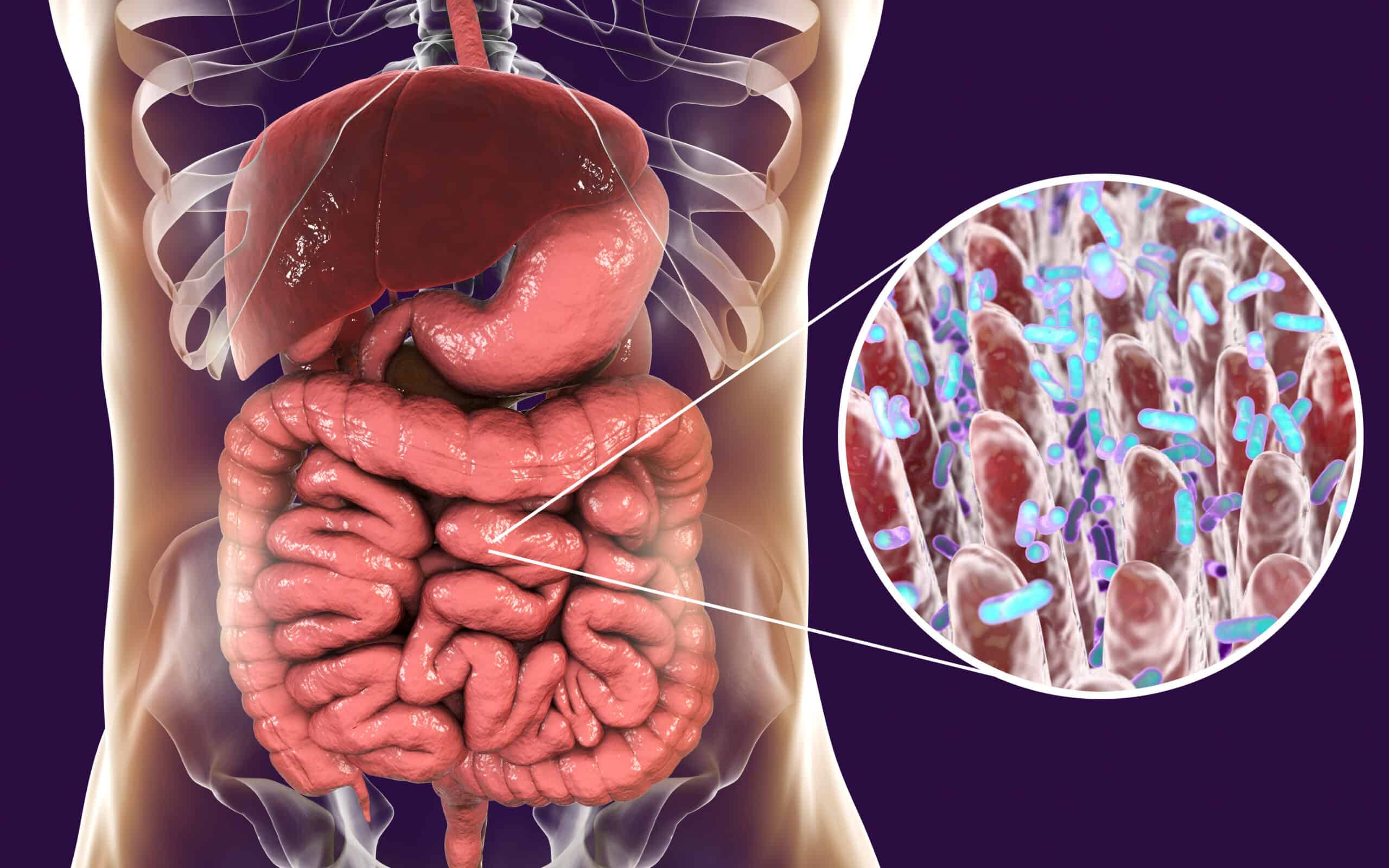
Microbes in your gastrointestinal tract make up your gut biome. The majority of these microorganisms consist of bacteria. Maintaining the good bacteria in your gut and minimizing bad bacteria is key to a healthy, well-functioning gut. The health of the gut also affects the brain and immune system. There is a very strong gut-brain connection, so what is happening in the gut also affects the brain (mood and cognition). In addition, a huge proportion — approximately 70% — of our immune system is in our GI tract.
Continue reading “Best Nutrients for Good Gut Health”Category: Gut Microbiota
The Many Benefits of a Medically Supervised Detox
Is something just not right? You want to feel your best, but vague conditions nag at you. Our bodies are exposed to toxins every day, through food, air or skin. This exposure takes a toll. Board-certified Gastroenterologist Dr. Lynne Ahn of Ahn Point Wellness discusses how a medically supervised detox enhances you physically and mentally.
Continue reading “The Many Benefits of a Medically Supervised Detox”Covid-19 and your Microbiome: It’s all about the GUT…
Increasing evidence shows the importance and impact of the gut microbiota on health and disease. There are clear associations of the gut microbial environment with intestinal infections, C. difficile, irritable bowel syndrome, inflammatory bowel disease, obesity, allergic diseases and neuropsychiatric disorders.
In a new observational study in Gut, by Yeoh and colleagues, correlation between gut microbiota composition, cytokine levels and inflammatory markers among COVID-19 patients demonstrated that the gut microbiome is linked with the severity of COVID-19. Gut microbiota alterations in association with immune dysregulation revealed that gut microorganisms are likely involved in the modulation of host inflammatory responses in COVID-19. Results showed gut microbiome changed among patients with COVID-19 vs. individuals without COVID-19 regardless of whether patients received medication (P < .01)
The authors show that selected gut commensals are depressed in the COVID-19 state compared with non-COVID controls. “Several gut commensals with known immunomodulatory potential such as Faecalibacterium prausnitzii, Eubacterium rectale and bifidobacteria were underrepresented in patients and remained low in samples collected up to 30 days after disease resolution,” Yeoh and colleagues wrote. “The dysbiotic gut microbiota that persists after disease resolution could be a factor in developing persistent symptoms and/or multisystem inflammation syndromes that occur in some patients after clearing the virus,” the authors wrote.
With mounting evidence that gut microorganisms are linked with inflammatory diseases within and beyond the gut, these findings demonstrate an urgent need to understand the specific roles of gut microorganisms in human immune function and systemic inflammation. The article by Yeoh et al is an interesting and compelling discussion of how the gut microbiota may have an impact on this disease state. “Bolstering of beneficial gut species depleted in COVID-19 could serve as a novel avenue to mitigate severe disease, underscoring importance of managing patients’ gut microbiota during and after COVID-19.”
Yeoh YK, Zuo T, Lui GC, et al
Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19
Gut 2021;70:698-706.